Imricor’s VISABL-AFL Trial for US FDA Approval Commences at Cardiovascular Institute of South Paris

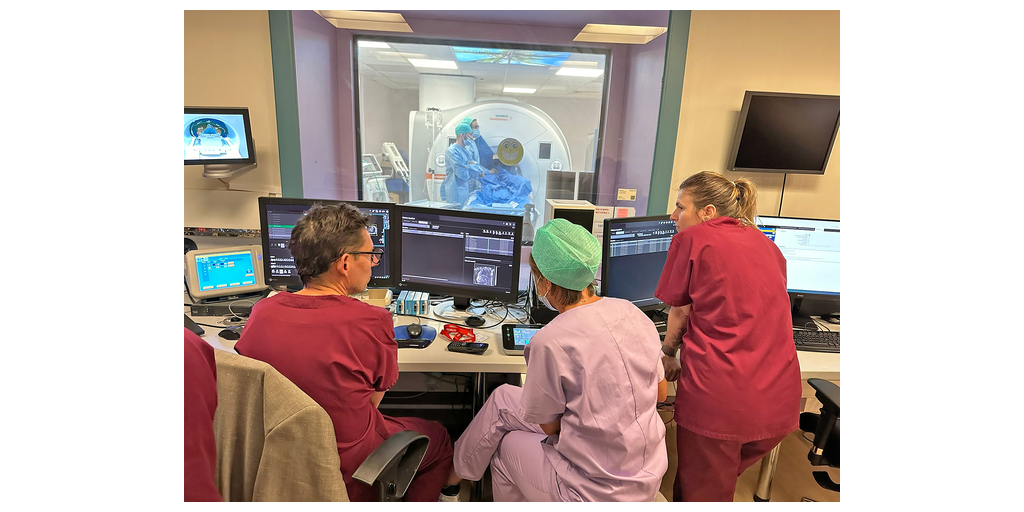
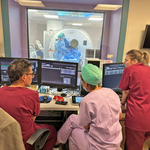
MINNEAPOLIS–(BUSINESS WIRE)–#CardioTwitter—Imricor Medical Systems, Inc. (Company or Imricor) (ASX:IMR) the global leader in real-time iCMR cardiac ablation products, is pleased to announce that the Vision-MR Ablation Catheter 2.0 for Treatment of Type I Atrial Flutter (VISABL-AFL) IDE clinical trial, commenced with two procedures performed at the Cardiovascular Institute of South Paris (ICPS) (https://icps.fr).
Dr. Laurent Fiorina, the operating Electrophysiologist at ICPS and the site’s Principal Investigator commented: “Enrolling the first patients in the VISABL-AFL clinical trial represents a significant step forward for the future of 3D real-time MRI ablations in the iCMR lab. Performing procedures with Imricor’s NorthStar 3D Mapping System is a game-changer for this field, and it will have a transformative impact. I look forward to the continued partnership with Imricor.”
The VISABL-AFL (NCT05904548) clinical investigation is a prospective, single-arm, multi-center, interventional, Investigational Device Exemption (IDE) trial. The primary objectives of VISABL-AFL are to assess the safety and efficacy of radiofrequency (RF) ablation of type-I atrial flutter performed with the Vision-MR Ablation Catheter 2.0 in the iCMR environment. The sample size is 91 patients, with an interim analysis after 76 patients have achieved the 7-day follow-up. The study includes four hospitals in the U.S. and Europe.
Professor Dr. Jerome Garot, Head of Cardiovascular Magnetic Resonance, commented: “We did the first two cases of RF flutter ablations with the Imricor system, and it went quite smoothly. We are very enthusiastic about it!”
Included in the VISABL-AFL trial is Imricor’s proprietary NorthStar Mapping System. NorthStar allows the user to control the MRI scanner, receive MR images in real-time from the scanner, display those MR images in 3D, actively track Imricor trackable devices, and create 3D electroanatomical maps that include intracardiac electrogram signals from Imricor’s Advantage-MR system.
Imricor’s Senior Director of Clinical, Kate Lindborg, commented: “Today’s important milestone would not have been possible without the partnership of the entire team at ICPS, whose collaboration and support are essential to the success of this trial. It is a privilege to work alongside such an esteemed hospital and the dedicated team at Imricor where we both share in the goal of improving patient outcomes and enhancing the quality of care.”
https://imricor.com/investors/about-imricor/
Contacts
Media Contact:
Nick Twohy
nick.twohy@imricor.com
1 952 818 8407
Recent Posts
Melco continues to lead with the city’s top number of Stars granted by MICHELIN Guide Hong Kong & Macau 2025
MACAO SAR - Media OutReach Newswire - 13 March 2025 - Melco Resorts & Entertainment…
Galaxy Macau’s Reveals a Galaxy of Stars As the Integrated Resort’s Restaurants are Awarded Five Michelin Stars in the 17th Michelin Guide Hong Kong Macau 2025
Sushi Kissho by Miyakawa Celebrates a Stellar Debut MACAU SAR - Media OutReach Newswire -…
Prince Holding Group Earns Fifth Consecutive ‘Best in Cambodia’ Award for CSR Initiatives
Recognized at the 17th Annual Global CSR & ESG Awards for Sustainable Development Efforts PHNOM…
LFC and adidas reunite with new multi-year partnership
LIVERPOOL, UK - Media OutReach Newswire - 13 March 2025 - Liverpool Football Club and…
Yuexiu Transport recorded net profit of RMB 660 million with stable final dividend of 13 HK cents per share
HONG KONG SAR - Media OutReach Newswire - 13 March 2025 - Yuexiu Transport (01052)…
VinUniversity and NTU Singapore to establish a strategic, comprehensive and long-term alliance
HANOI, VIETNAM - Media OutReach Newswire - 13 March 2025 - VinUniversity (VinUni) and Nanyang…